By: Alex Feldmeyer & Abbey Thiel
Dalgona coffee first gathered attention in South Korea among social media platforms, namely Tiktok. Eventually it made its way to other countries and assumed the same popularity. At this point, many of you have either tried this whipped coffee drink or know someone who has. At the very least, you’ve probably seen a picture on your phone or computer! It’s amazing how fast trends sweep the world with a little more time on our hands.
More time at home can translate to boredom, but often it also makes people more creative and willing to try something new. There have probably been more loaves of bread baked in the last 3 months than ever before… According to a NY Times briefing, “King Arthur’s grocery-store sales rose 600 percent almost overnight“.
The whipped coffee taking the internet by storm is called Dalgona coffee. “Dalgona” actually references a Korean honeycomb toffee with a similar flavor.

Photo by Oana Cristina on Unsplash
As with most food and beverage trends, the science of their allure is just as enticing as the actual consumption. (Well at least for some of us…!)
If you haven’t tried this treat yet, here is a simple recipe.
Ingredients for Dalgona Coffee:
In equal parts, whip
- Instant coffee
- Sugar
- Water
Once frothy, you can pour on top of a milk/dairy alternative base of your choice.
Interestingly enough, Dalgona coffee can only be made from instant coffee – fresh coffee grounds will not form the luxurious froth. But before we discuss the theory as to why this sensation is specific to instant coffee, let’s talk about another aspect of the drink: foam.
Foam formation
Although the texture of this beverage varies from traditional morning Joe foams, they are not a foreign concept when it comes to coffee. The “crema” on top of a properly prepared espresso, is in fact considered a foam.

Photo by Nathan Dumlao on Unsplash
A foam is defined as coarse dispersion of gas bubbles (carbon dioxide generated from coffee roasting) in a liquid continuous phase (oil droplets in watery mixture of sugar, acids, proteins and caffeine)1. The “crema” foam is characteristic of an espresso, consumed religiously in Italy and much of Europe. The ideal crema is about 10% of the espresso serving.

Image by Abbey Thiel4
There appears to be a relationship between foam stability and high molecular weight sugars, as well as some of the coffee roasting by-products: melanoidins. Melanoidins are strings of nitrogen-containing molecules derived by the roasting process that impart taste, aroma and apparently foam formation1-2. These compounds may help lower the interfacial tension between air bubbles and the coffee phase, aiding in foam formation and stabilization, as impurities have been suggested to do.
The reason why the combination of instant coffee, sugar and water seems to magically create a velvety frothy masterpiece is actually a little bit elusive. This is different from espresso foams in that it doesn’t involve pulling espresso or brewing the coffee itself. It relies on the functionality of instant coffee – which can be produced via spray-dry or freeze-dry techniques.
Instant Coffee
The first step in instant coffee production consists of roasting coffee beans, grinding them, and brewing3.
For Spray drying, the concentrated liquid coffee is “misted” into hot, dry air (≈480°F). At this temperature the residual water evaporates and the coffee particles dry up, forming small particulates.
Freeze-drying involves concentrating the coffee extract and then chilling it. It’s further cooled to -40°F and forms ice. This ice is broken down and then dried in a vacuum, vaporizing the ice and leaving the coffee particles3.
One theory as to why instant coffee is the key ingredient to this whipped beverage is that its production has made the particles easier to hydrate than coffee grounds. Additionally, the coffee has already been brewed, so the instant coffee contains the brewing by-products that enable foam formation (which would not be present in coffee grounds prior to their roast).
Sugar, another ingredient in this beverage, also aids in the foam-ability in a similar manner.
Sugar and its role in foam stabilization
Sugar adds viscosity, thickening the coffee phase. This makes these “liquids” flow a lot slower, while also potentially slowing the foam’s collapse, keeping your drink frothy longer!
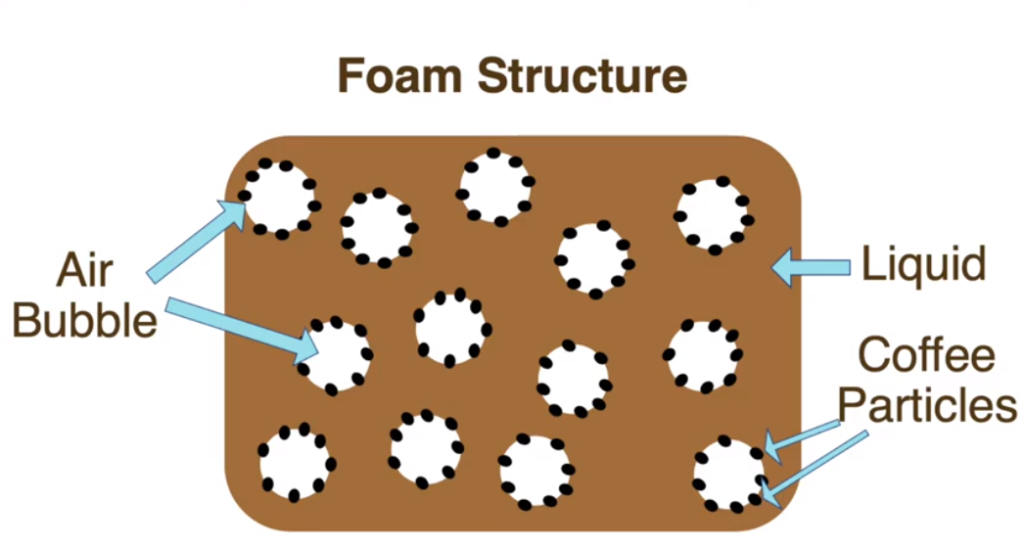
Image by Abbey Thiel4
Here’s to hoping we have demystified one of the quarantine’s biggest mysteries. Maybe you will feel inspired to create your own viral beverage? Only time will tell!
References
- Illy, E., & Navarini, L. (2011). Neglected food bubbles: the espresso coffee foam. Food biophysics, 6(3), 335-348.
- Rufián-Henares, J. A., & Pastoriza, S. (2015). Melanoidins in coffee. In Coffee in Health and Disease Prevention(pp. 183-188). Academic Press.
- https://www.huffpost.com/entry/what-is-instant-coffee_n_57bda1ebe4b00d9c3a1afc74
- https://www.youtube.com/watch?v=5_ju1nvS9fU&t=238s
- Featured Image: Image by ivabalk from Pixabay
 Alex Pierce-Feldmeyer | Linkedin | Website
Alex Pierce-Feldmeyer | Linkedin | Website
Alex graduated with a B.S. from the University of Illinois in Food Science and completed her PhD at Ohio State University in a lab revolving around sensory evaluation and psychophysics, potentially the functional or cognitive benefits from food and food ingredients. Alex is currently a sensory scientist at MANE, a flavor company. An avid study-er of the nose and its wild contributions to flavor, potentially how aroma affects different aspects of cognition. Alex dabbles in triathlons and fitness-ness. She has an ice cream tour every Sunday, every ice cream place (that is good) is on her list and it must have sprinkles. Grocery stores, sunrises and puppies are everything!

Abbey Thiel | Linkedin | YouTube | Medium
Abbey discovered that food science was a real major while attending University of Wisconsin-Madison, and quickly transferred into the department. After graduating with a B.S. in food science, she decided to stay in Madison to pursue her PhD. Her research explores the fat network found within ice cream, and yes, there’s lots of ice cream parties! If not found sitting at a microscope for extended amounts of time, Abbey enjoys the great Midwestern sport of log rolling (Google it) and laughing really loud. She is also very busy passing on her passion for food science to her two-year-old niece, whose favorite color is bacon and has hopes of growing up to be soup.






Leave a Reply